There are several factors that are used to determine the state of the industry, from its economic value and allocated governmental budgets to the development of technologies reshaping treatment
As with many industries, the two core concepts driving medtech change are the IoT and digitalisation. These technologies present a wealth of new possibilities for the sector, but bring with them the challenges of equipment safety, power quality and system continuity.
Healthcare trends
Medtech is in a transformative period, driven by several technological and social trends. These range from changes in the population to the rise of digital healthcare technology.
There are five key trends that are reshaping the industry.
Home and mobile healthcare
With the rise of digital healthcare technology has come the increased use of healthcare apps and devices in the home. Much of the home healthcare or mobile health (mHealth) trend is being driven by the development of smart phone technology allowing health apps to provide more comprehensive analysis of user conditions, as well as the advancement and popularity of consumer wearable electronics.
These applications currently offer limited insight into user health, but a 2014 report by Deloitte states, “the potential lies in supporting higher-impact clinical decision-making and developing the interaction between clinicians and patients”.
Rather than signalling the commoditisation of healthcare, the rise of mHealth has triggered a shift in how the UK’s healthcare industry operates. In 2015, the NHS announced proposals to develop the capability for mHealth applications and wearables to sync directly with NHS patient records.
In 2017, the NHS shared its digital apps library. This is a directory of NHS-certified healthcare applications that, along with a companion site to help app developers meet NHS guidelines, helps patients identify and use trustworthy apps.
A significant factor in the development of mHealth technology has been the financial pressures and time constraints faced by healthcare practitioners.
Digitisation
The NHS announced that it was going to trial the use of apps for patient care in four NHS trusts by early 2018. Clinical trials of the apps showed a reduction in hospital admissions and GP appointments. The use of mHealth technology will continue to increase in coming years, in both home and hospital environments.
The ageing population
As we age, we become more susceptible to illness and infection, therefore an ageing population inevitably increases the volume of hospital admissions and practitioner appointments, placing significant pressure on healthcare systems.

New medtech devices and equipment must be able to operate reliably and safely, both functionally and practically. For example, many pieces of equipment must be washed down properly between uses to reduce the risk of infection to elderly patients, so devices must be designed accordingly.
Wearable medtech has contributed to addressing the issues that come with an ageing population, allowing home healthcare devices to monitor patients and ensure medical staff are notified if anything requires urgent attention.
The ageing and less mobile population seemingly drives a need for devices that are mobile. With this comes a paradigm shift for the healthcare sector, from reactive care of illness to proactive maintenance of health. Digital medtech manufacturers will have to find ways of designing this approach into their products, as well as ensuring any reactive elements — such as emergency staff responding to sudden changes in a patient at home — are acted on promptly.
Medical data security
The rise of medical devices that monitor, record and process patient data might mean better and more comprehensive care, but it also exposes the patient to risk.
In 2017, the WannaCry ransomware attack on NHS systems resulted in disruption and expense in cancelled appointments, IT support and the cost of system restoration.
Many, including the NHS’s deputy chief executive Rob Shaw, believe these attacks will continue. Shaw has stressed that “the threats we have seen during 2017 are unlikely to be going anywhere, with low-effort, high reward ransomware continuing to be prevalent along with more sophisticated command and control structures, making it more difficult to employ traditional mitigation and remediation methods”.
While WannaCry was the most high profile, it was not the only threat to NHS systems or patient data. There is a general threat to system continuity through the reliability of IT hardware.
Many IT systems used by NHS hospitals are outdated, which was a contributing factor in the WannaCry infiltration. These systems will also have inefficient hardware that can contribute to EMI and can therefore make medical equipment unreliable.
It is essential that healthcare organisations do not improve cybersecurity procedures yet neglect the performance of the hardware used to access the data in the process.
Portable devices and complex setups
Particularly since the advent of the IoT in healthcare environments, the number of devices in a typical hospital has significantly increased. The rising use of portable and handheld devices, including vital patient monitoring and analysis devices as well as endoscopy equipment and tablet computers, has changed the face of healthcare.
Mainstays have also been replaced with a digital or electronic alternative. Notepads have been replaced with iPads, mechanical beds with electrically adjustable ones and traditional film imaging has been substituted for the digital picture archiving and communication system (PACS).
The increasing electrification of devices creates problems on the mains power supply. AC to DC power conversion by the device’s power supply introduces issues such as voltage distortion, current harmonics, ESD, power surges and EMI into the supply.
These power quality problems can affect the calibration and sensitivity of diagnostic devices used by doctors and healthcare professionals. Erroneous test results can result in misdiagnosis and potentially harmful treatment plans for patients.
That comes without considering the consumer devices that are increasingly common in hospitals. Commercial off-the-shelf (COTS) equipment is often used in hospitals alongside medical equipment. Equipment, such as commercial PCs, can result in electrical interference and have serious effects on medical equipment functionality.
For example, there have been reports of cases where critical medical equipment such as defibrillators failed to work because of interference from secondary equipment, such as ambulance radios.
As digitalisation brings new benefits to healthcare, engineers must be primed to eliminate the problematic by-products of these developments. Design engineers must ensure devices are designed to comply with electrical standards, such as EMC directives to minimise EMI and systems must be kept safe from power quality problems.
Surgical robots
According to a report from market research firm Allied Market Research, the global surgical robotics market will be valued at $28.8bn by 2020.
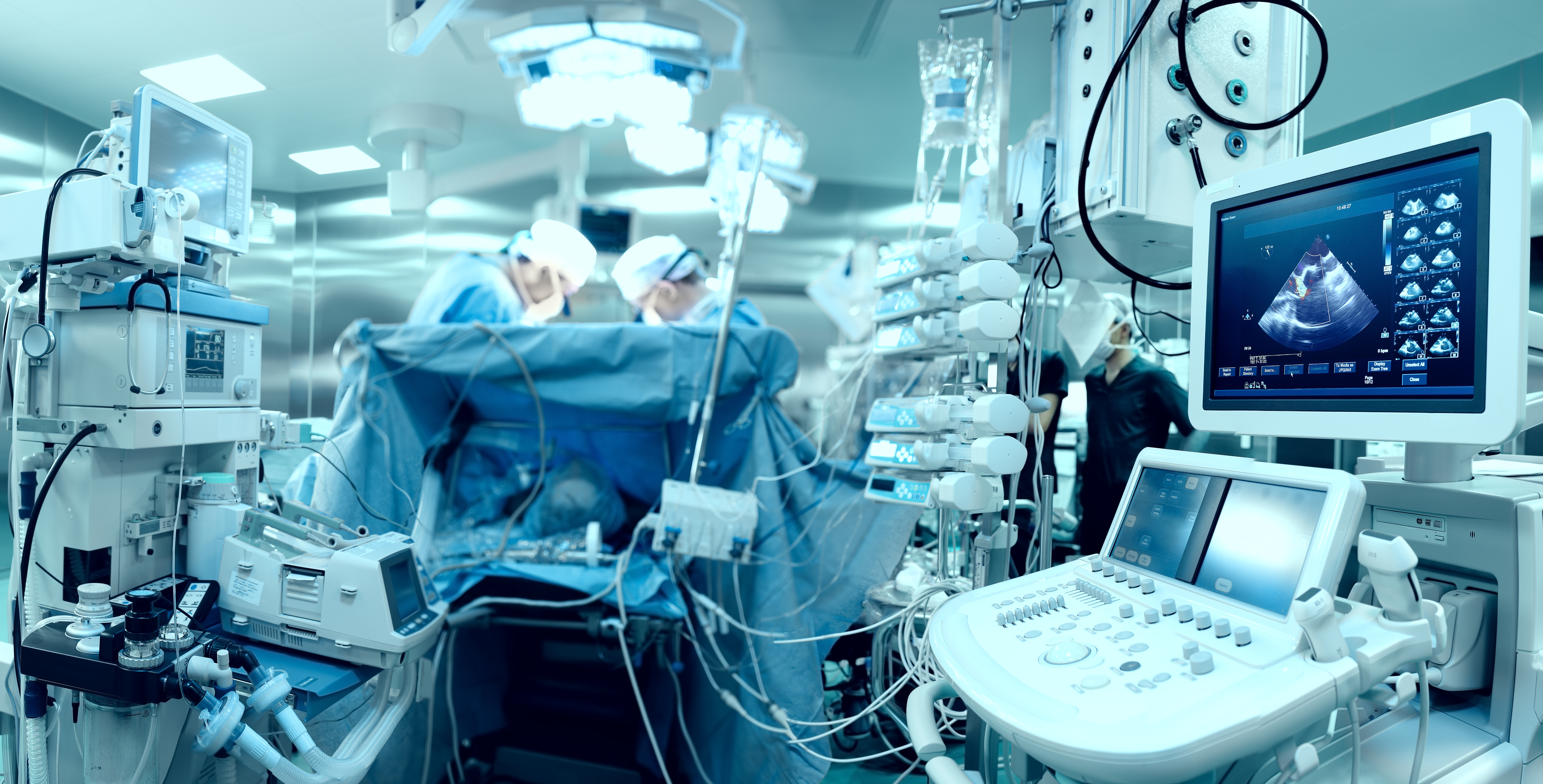
There are several reasons why surgical robots are becoming increasingly prevalent. One of the biggest is that collaborative robotics allows surgeons to work for longer without the health issues, such as back pain during long surgical procedures.
The other argument for surgical robots is that they greatly enhance accuracy during keyhole surgery. According to Graham Mackrell, managing director of robot gear manufacturer, Harmonic Drive UK, “Surgical robots used for keyhole surgery allow surgeons to be more precise in their incisions as the system handles the multiple apparatus required. Instead of a surgeon effectively juggling the laparoscope and surgical tools, the robot can handle both simultaneously for a quicker and more efficient operation”.
A designer’s dilemma
Safety is critical when designing medical equipment; its importance is underlined by the wide range of regulations and directives relating to medical equipment, such as the European EN60601-1 standard, which governs the safety, essential performance and EMC of equipment.
The directives classify devices into three areas based on how closely they are used to the patient’s body. Type B (body) devices operate within a six-foot vicinity of the patient without bodily contact. Type BF (body floating) makes physical contact with the body and Type CF (cardiac floating) makes physical contact with the heart. Each of the categories outlines the level of isolation, insulation, creepage, clearance and leakage allowed.
The EU has introduced several new regulations and directives in the past few years that affect how medical devices are designed, developed and manufactured. The medical devices regulation (MDR 2017/745) was published in mid-2017 and will be in effect from mid-2020 onwards.
The MDR details an increased focus on product lifecycle requirements rather than solely pre-approval processes, with the aim to make medical devices consistently safer from cradle to grave. In addition to this, EN ISO 13485:2016 is mandatory as of 2019 and will put even greater focus on quality management and risk management as part of product development.
More regulation means more headaches for design engineers, but the greater focus on safety and reliability across the entire product lifecycle offers an opportunity for design engineers to positively impact the wider industry. By keeping power considerations in mind during the design stages and being mindful of EMC directives, design engineers can keep devices compliant while maintaining clean power in healthcare environments.
Resistive and inductive wound components are used in the REOMED range of isolating medical transformers, which can be integrated into medical devices to comply with EMC directives as well as EN60601.
The transformers allow safe galvanic separation between the primary and secondary electrical circuits, which limits the electrical leakage and, as such, the interference to other devices. It also keeps the equipment safe from poor power quality introduced to a network by mains-connected consumer devices.
Isolating transformers are often embedded in large clinical equipment, such as MRI machines and anaesthesia units, to maximise safety. Equipment for patient environments, such as operating theatres, however, does not often contain an embedded transformer to isolate the unit. Introducing one into the design stages could offer greater safety over the course of the product lifecycle, complying with the increasingly stringent EU directives
The rise in portable and handheld devices used to monitor patient vitals increases the number of devices in hospitals and, with more devices used, the risk of electrical interference rises. Such devices are susceptible to changes in power quality, which affects the quality of data received.
These changes in power quality can lead to issues like EMI, which causes surges and spikes in power. This has an impact on both the data collected by medical equipment and the way devices interact with each other, as well as staff access to patient records.
If interference creates an unreliable power supply to IT systems or a damaging power surge, medical staff may be unable to access these files, yet such events can be mitigated by using network isolators and isolation monitors. In the same way that an isolating transformer separates a piece of equipment from mains power, network isolators such as the REOMED Isonet, provide effective electrical isolation of devices in copper wire-bound Ethernet networks. The Isonet also protects both equipment and people from the impact of electrical voltage spikes.
In other medical environments such as operating theatres, isolating transformers can be used to ensure critical equipment has galvanic separation from the mains to ensure a consistent and reliable performance.
Depending on the transformer used, this can also reduce operating costs. Traditional medical transformers typically have quite high electrical losses, which results in higher energy costs and a greater environmental impact. Toroidal transformers have been tested and consistently shown to deliver significantly lower losses. Where a conventional transformer with a capacity of 2,200VA might exhibit losses of 1,13W during use, a REOMED transformer with the same capacity would typically exhibit losses of 62.5W, a 45% reduction.
Reliable power is just as central to medical and healthcare environments as those industries are to our society. For all the challenges facing the industry, the first step to overcoming them is to ensure the equipment and networks can facilitate the future solution. Good power quality is critical in ensuring this in an efficient, safe and reliable way.









